When the index n is between any two of the former values (0, 1, γ, or ∞), it means that the polytropic curve will cut through (be bounded by) the curves of the two bounding indices. Under the assumption of ideal gas law, heat and work flows go in the same direction ( K < 0), such as in an internal combustion engine during the power stroke, where heat is lost from the hot combustion products, through the cylinder walls, to the cooler surroundings, at the same time as those hot combustion products push on the piston.Įquivalent to an isochoric process (constant volume) N 0), such as in vapor compression refrigeration during compression, where the elevated vapour temperature resulting from the work done by the compressor on the vapour leads to some heat loss from the vapour to the cooler surroundings.Įquivalent to an isentropic process (adiabatic and reversible, no heat transfer), under the assumption of ideal gas law. 
Variation of polytropic index n Polytropic 
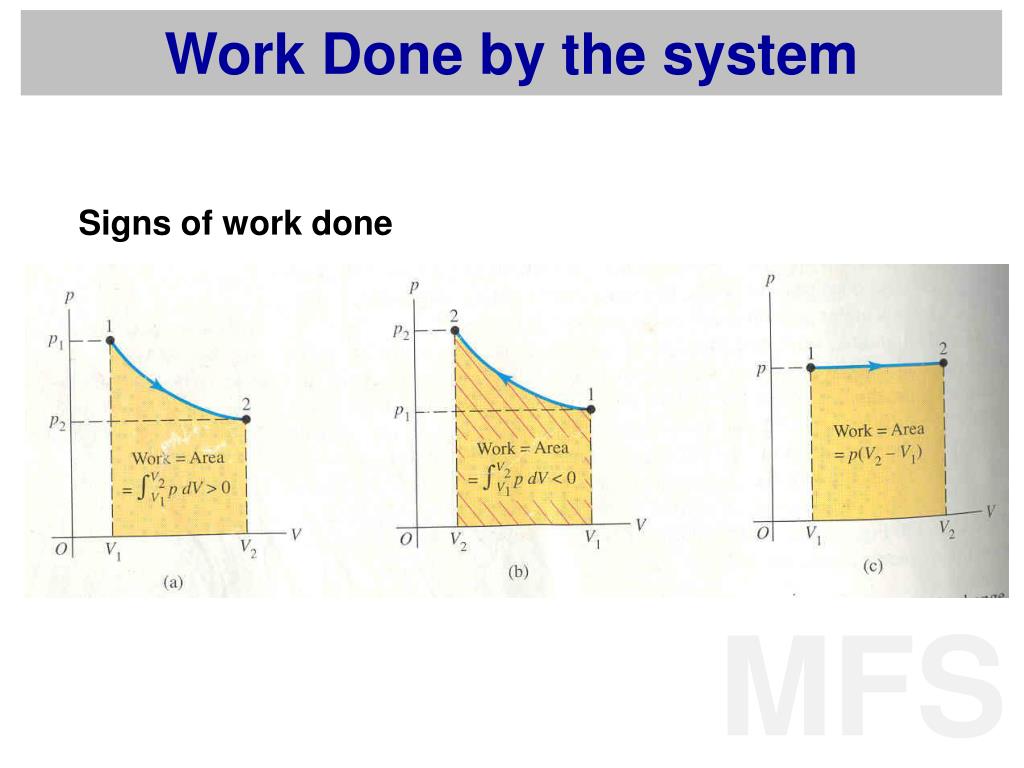
e consider the work done by the system during a. Some examples of the effects of varying index values are given in the following table. of the great power and generality of thermodynamics is that it does not details of the structure of matter. Heat engines, heat pumps, refrigerators, and air conditioners are examples of such systems. Look for any system where the primary methods of transferring energy are heat and work. įor certain values of the polytropic index, the process will be synonymous with other common processes. PROBLEM SOLVING STRATEGIES FOR THERMODYNAMICS Examine the situation to determine whether heat, work, or internal energy are involved.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
